Device-Enhanced Delivery for the Benefit of Patients
A key aim of the Oxford Centre for Drug Delivery Devices is to faciliate the clinical translation of physical methods of enhanced drug delivery, in order to enable current and future drugs to fullfil their therapeutic potential.
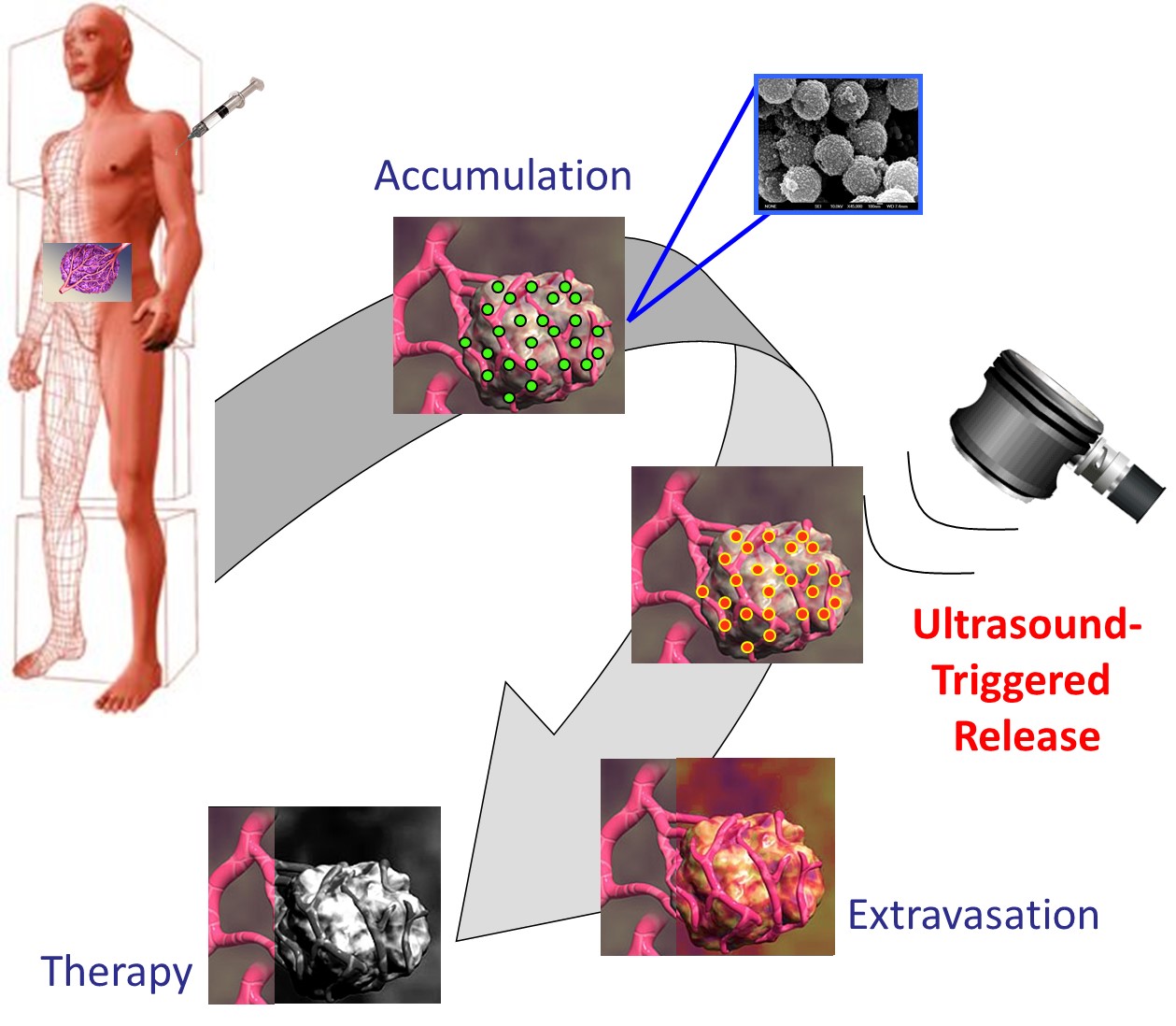
In partnership with the Oxford Biomedical Research Centre, a first-in-man trial of ultrasound-enhanced drug delivery for cancer received ethics approval (clinicaltrials.gov NCT02181075) and has now been initiated. As a Phase I trial, TarDox seeks to demonstrate the safety and feasibility of triggered release and enhanced delivery by ultrasound-mediated mild hyperthermia of doxorubicin from thermosensitive liposomes in patients suffering from primary or metastatic cancers in the liver. The trial is currently recruiting and is expected to report towards the end of 2017.